Adjuvants in Microencapsulation-Based Vaccines for Cantang Grouper against Viral Nervous Necrosis: A Review
Keywords:
chitosan, Freund’s adjuvant (CFA/IFA), microencapsulation, nanovaccine, viral nervous necrosis, aquacultureAbstract
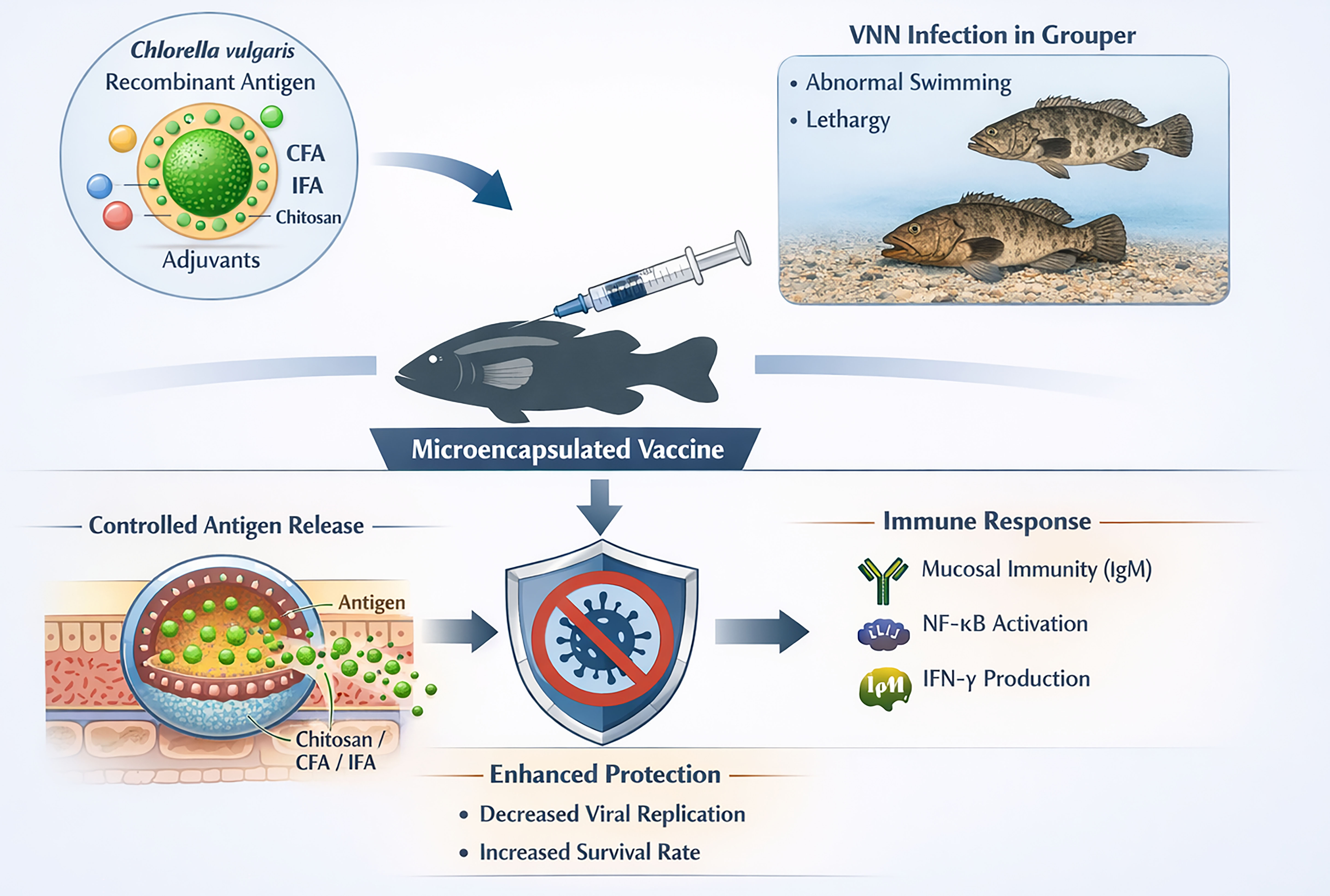
Viral nervous necrosis (VNN) is one of the most serious diseases attaching Cantang grouper (Epinephelus fuscoguttatus × Epinephelus lanceolatus) with mortality up to 100% in juvenile fish in aquaculture. A promising strategy to control this disease is the development of recombinant vaccines using the microalgae Chlorella vulgaris. In vaccine formulations, the addition of adjuvants is important for enhancing fish immune responses. This study aims to review the microencapsulation techniques using recombinant C.vulgaris protein combined with adjuvants such as Complete Freund’s Adjuvant (CFA), Incomplete Freund’s Adjuvant (IFA), and chitosan to improve immune responses in Cantang grouper infected with VNN. This review was conducted by synthesizing peer-reviewed journals published over the last decade. The analysis focuses on evaluating the integration of various adjuvants with microencapsulation technologies to enhance vaccine efficacy against VNN in Cantang Grouper. The results indicate that the use of recombinant C.vulgaris-based vaccines combined with encapsulated adjuvants, particularly CFA/IFA and chitosan, can enhance immune responses in Cantang grouper. Microencapsulation technology enhances antigen stability and enables controlled antigen release, thereby improving vaccine delivery efficiency. These findings suggest that adjuvant-assisted microencapsulation systems have significant potential to improve vaccine effectiveness to prevent fish from VNN infection in aquaculture. Technological applications should focus on optimizing encapsulation formulas and large-scale trials to ensure long-term immunity in sustainable grouper farming.
References
[1] Yanuhar U, Waheed Z, Suryanto H, Hasan V, Machfuda DR. Efficacy of zinc oxide chitosan hybrid nanoparticles as a nanovaccine to enhance immunity in hybrid grouper fish. Global J Environ Sci Manage 2026; 12:1–18. https://doi.org/10.22034/gjesm.2026.01
[2] Junirahma NS, Yanuhar U. Immune response of VNN (viral nervous necrosis) infected grouper utilizing Chlorella vulgaris extract as an anti-virus candidate. J Phys Conf Ser 2020; 1665: 012008. https://doi.org/10.1088/1742-6596/1665/1/012008
[3] Wu Y-C, Hong J-R, Chi S-C. Nodaviruses of fish. In: Kibenge FSB, Godoy MG (eds). Aquaculture virology (second edition), London: Academic Press; 2024, p. 515–43. https://doi.org/10.1016/B978-0-323-91169-6.00019-4
[4] Hao K, Wang Y, Zhu B, Yu F, Zhao Z, Wang G-X. Recombinant surface display vaccine enhances the immersion immune effect against grass carp reovirus in grass carp (Ctenopharyngodon idella). Fish Shellfish Immunol 2023;142:109160. https://doi.org/10.1016/j.fsi.2023.109160
[5] Cadar E, Popescu A, Dragan A-M-L, Pesterau A-M, Pascale C, Anuta V, et al. Bioactive compounds of marine algae and their potential health and nutraceutical applications: A review. Mar Drugs 2025;23. https://doi.org/10.3390/md23040152
[6] Lalarukh, Hussain SM, Ali S, Yilmaz E, Zahoor AF, Javid A, et al. Microencapsulation: An innovative technology in modern science. Polym Adv Technol 2025;36: e70066. https://doi.org/10.1002/pat.70066
[7] Correa VA, Portilho AI, De Gaspari E. Vaccines, adjuvants and key factors for mucosal immune response. Immunology 2022; 167:124–38. https://doi.org/10.1111/imm.13526
[8] Shams N, Jaydari A, Najafi H, Hataminejad M, Khanizadeh S, Pouladi I. An overview of the types of adjuvants used in the vaccination industry and their mechanisms of action. Viral Immunol 2024; 37:324–36. https://doi.org/10.1089/vim.2024.0032
[9] Ou B, Yang Y, Lv H, Lin X, Zhang M. Current progress and challenges in the study of adjuvants for oral vaccines. BioDrugs 2023; 37:143–80. https://doi.org/10.1007/s40259-022-00575-1
[10] Parmaksız S, Şenel S. An overview on Chitosan-based adjuvant/vaccine delivery systems. In: Jayakumar R, Prabaharan M, editors. Chitosan for biomaterials IV: biomedical applications, Cham: Springer International Publishing; 2021. p.293–379. https://doi.org/10.1007/12_2021_93
[11] Astari B, Effendi I, Budiardi T, Hadiroseyani Y, Diatin I, Ismi S. Financial analysis business of Cantang (Epinephelus fuscoguttatus × E. lanceolatus) and Cantik (E. fuscoguttatus × E. polyphekadion) hybrid groupers in hatcheries. Aquac Rep 2024; 39:102364. https://doi.org/10.1016/j.aqrep.2024.102364
[12] Haini N, Sumsanto M. Study of hybrid Cantang grouper breeding techniques (Epinephelus fuscoguttatus x Epinephelus lanceolatus) at the brackish water fisheries cultivation center Situbondo, East Java. J Fish Health 2025; 4:142–56. https://doi.org/10.29303/jfh.v4i3.5933
[13] Nadia LOAR, Kasim R, Ramli M, Yusnaini Y. Diversity and characteristics of the habitat of groupers (genus Epinephelus) in Moramo Bay, Southeast Sulawesi, Indonesia. AACL Bioflux 2024; 17:1170–7. https://bioflux.com.ro/docs/2024.1170-1177.pdf
[14] Okon EM, Oyesiji AA, Okeleye ED, Kanonuhwa M, Khalifa NE, Eissa E-SH, et al. The escalating threat of climate change-driven diseases in fish: evidence from a global perspective – a literature review. Environ Res 2024; 263:120184. https://doi.org/10.1016/j.envres.2024.120184
[15] Chen C, Ward VCA. Recombinant protein expression and its biotechnological applications in Chlorella spp. SynBio 2024; 2:223–39. https://doi.org/10.3390/synbio2020013
[16] Yanuhar U, Mahasri G, Suryanto H, Syamlan SBY, Junirahma NS, Caesar NR. The effect of Chitosan hybrid nanoparticle-based nanovaccines (C-Nps) on the tissue damage of cantang grouper fish infected with viral nervous necrosis. AIP Conf Proc 2024; 3027:020011. https://doi.org/10.1063/5.0205069
[17] Zorriehzahra MJ. Viral nervous necrosis disease. In: Ennaji MM, editor. Emerging and reemerging viral pathogens. London: Academic Press; 2020. p. 673–703. https://doi.org/10.1016/B978-0-12-819400-3.00030-2
[18] Nurlita W, Pandit IGS, Darmadi NM. Detection of the existence of viral nervous necrosis on fry Cantang grouper at rain season. Sustain Environ Agric Sci 2020; 4:46–52. https://doi.org/10.22225/seas.4.1.1687.46-52
[19] Nwaubani NA, Sherchan SP. The impact of climate change on the transmission of enteric diseases: a systematic review. Tot Environ Microbiol 2026; 2:100056. https://doi.org/10.1016/j.temicr.2026.100056
[20] Ariff N, Abdullah A, Azmai MNA, Musa N, Zainathan SC. Risk factors associated with viral nervous necrosis in hybrid groupers in Malaysia and the high similarity of its causative agent nervous necrosis virus to reassortant red-spotted grouper nervous necrosis virus/striped jack nervous necrosis virus strains. Vet World 2019; 12:1273–84. https://doi.org/10.14202/vetworld.2019.1273-1284
[21] Zhiqi Z, Jing X, Xiaoqian T, Xiuzhen S, Heng C, Wenbin Z. Identification of B-cell epitopes on capsid protein reveals two potential neutralization mechanisms in red-spotted grouper nervous necrosis virus. J Virol 2023; 97:e01748-22. https://doi.org/10.1128/jvi.01748-22
[22] Yanuhar U, Arfiati D, Musa M, Kusriani, Junirahma NS, Caesar NR. The status of VNN (viral nervous necrosis)-infected grouper fish tissue with Chlorella vulgaris extract as anti-virus candidate. J Phys Conf Ser 2020; 1665:012036. https://doi.org/10.1088/1742-6596/1665/1/012036
[23] John KR, Sivasankar P. George MR. Status of emerging and reemerging fish viral infections in India. In: Saxena SK, editor. Viral outbreaks - global impact and newer horizons, London: IntechOpen; 2023. https://doi.org/10.5772/intechopen.109012
[24] Aich N, Paul A, Choudhury TG, Saha H. Tilapia lake virus (TiLV) disease: current status of understanding. Aquac Fish 2022; 7:7–17. https://doi.org/10.1016/j.aaf.2021.04.007
[25] Wang C, Liu S, Xu T, Li X, Li J, Zhang Q. Pathogenicity study of covert mortality nodavirus (CMNV) infection in zebrafish model. Aquaculture 2022; 546:737378. https://doi.org/10.1016/j.aquaculture.2021.737378
[26] Ru ITK, Sung YY, Jusoh M, Wahid MEA, Nagappan T. Chlorella vulgaris: a perspective on its potential for combining high biomass with high value bioproducts. Appl Phycol 2020; 1:2–11. https://doi.org/10.1080/26388081.2020.1715256
[27] Fučíková K, Pažoutová M, Rindi F. Meiotic genes and sexual reproduction in the green algal class Trebouxiophyceae (Chlorophyta). J Phycol 2015; 51:419-430. https://doi.org/10.1111/jpy.12293
[28] Yanuhar U, Suryanto H, Mahasri G, Rahman Caesar N. Cloning and characterization of protein fraction specific of Chlorella vulgaris endemic East Java. Evergreen 2024; 11:82–94. https://doi.org/10.5109/7172229
[29] Arguelles DLRE. Morphotaxonomic Study of algal epiphytes from Ipomoea aquatica Forssk. (Convolvulaceae) found in Laguna de Bay (Philippines). Pertanika J Trop Agric Sc 2019; 42:817–32. http://www.pertanika.upm.edu.my/pjtas/browse/regular-issue?article=JTAS-1496-2018
[30] Zullaikah S, Jessinia MCP, Rinaldi R, Yasmin M, Rachimoellah M, Wu DW. Lipids extraction from wet and unbroken microalgae Chlorella vulgaris using subcritical water. Mater Sci Forum 2019; 964:103–8. https://doi.org/10.4028/www.scientific.net/MSF.964.103
[31] Coronado-Reyes JA, Salazar-Torres JA, Juárez-Campos B, González-Hernández JC. Chlorella vulgaris, a microalga important to be used in Biotechnology: a review. Food Sci Technol (Brazil) 2022;42. https://doi.org/10.1590/fst.37320
[32] Saidu I, Abu GO, Akaranta O, Chukwuma FO, Vijayalakshmi S, Ranjitha J. Biosynthesis and characterization of algae oil obtained from Chlorella vulgaris. J Mater Sci Res Rev 2021; 4:230–40. https://journaljmsrr.com/index.php/JMSRR/article/view/127
[33] Masitha A, Yanuhar U, Suryanto AMH. In-vivo test of Chlorella vulgaris extract for induction into heat shock proteins of Cantang grouper (Ephinephelus fuscoguttatus-lanceolatus) infected by viral nervous necrosis. J Fish Mar Res 2019; 3:22–31. https://doi.org/10.21776/ub.jfmr.2019.003.01.3
[34] Yanuhar U, Al Hamidy I, Caesar NR. Treatment of Chlorella sp. extract on heat shock cluster (HSC) response from the tissue and blood cells proliferation of Epinephelus fuscoguttatus-lanceolatus infected by viral nervous necrosis. IOP Conf Ser Earth Environ Sci 2019; 236:012100. https://doi.org/10.1088/1755-1315/236/1/012100
[35] Andriyani WM, Murtini S, Alimuddin A. Effication of viral nervous necrosis DNA as vaccines for Cromileptes altivelis. Indones J Trop Aquat 2020;3. https://doi.org/10.22219/ijota.v3i2.13891
[36] Facciolà A, Visalli G, Laganà A, Di Pietro A. An overview of vaccine adjuvants: current evidence and future perspectives. Vaccines (Basel) 2022;10. https://doi.org/10.3390/vaccines10050819
[37] Fontes JA, Barin JG, Talor M V, Stickel N, Schaub J, Rose NR, et al. Complete Freund’s adjuvant induces experimental autoimmune myocarditis by enhancing IL-6 production during initiation of the immune response. Immun Inflamm Dis 2017; 5:163–76. https://doi.org/10.1002/iid3.155
[38] Pollack KE, Meneveau MO, Melssen MM, Lynch KT, Koeppel AF, Young SJ, et al. Incomplete Freund's adjuvant reduces arginase and enhances Th1 dominance, TLR signaling and CD40 ligand expression in the vaccine site microenvironment. J Immunother Cancer 2020;8(1):e000544. https://doi.org/10.1136/jitc-2020-000544
[39] Huda C, Yanuhar U, Musa M, Wahyudi Y, Junirahma NS, Caesar N. Antibody response in the blood of Cantang grouper infected by viral nervous necrosis with recombinant protein Chlorella vulgaris vaccination. J Aquac Fish Health 2023; 12:144–54. https://doi.org/10.20473/jafh.v12i2.39231
[40] Lei Z, Langrish T. A Review of the extraction and closed-loop spray drying-assisted micro-encapsulation of algal lutein for functional food delivery. Processes 2021;9. https://doi.org/10.3390/pr9071143
[41] Angulo C, Tello-Olea M, Reyes-Becerril M, Monreal-Escalante E, Hernández-Adame L, Angulo M, et al. Developing oral nanovaccines for fish: a modern trend to fight infectious diseases. Rev Aquac 2021; 13:1172–92. https://doi.org/10.1111/raq.12518
[42] Sanina N. Vaccine adjuvants derived from marine organisms. Biomolecules 2019;9. https://doi.org/10.3390/biom9080340
[43] Brai A, Poggialini F, Pasqualini C, Trivisani CI, Vagaggini C, Dreassi E. Progress towards adjuvant development: focus on antiviral therapy. Int J Mol Sci 2023, 24, 9225. https://doi.org/10.3390/ijms24119225
[44] Rodríguez A, Gadan K, Pérez L, Evensen Ø, Estrada MP, Carpio Y. Prime-boost vaccination with chimeric antigens adjuvanted in MontanideTM ISA50 V2 confers protection against experimental Lepeophtheirus salmonis infestation in Atlantic salmon (Salmo salar L.). Front Immunol 2025;16. https://doi.org/10.3389/fimmu.2025.1570948
[45] Rizkiantino R, Wibawan IWT, Pasaribu FH, Soejoedono RD, Poetri ON, Arnafia W, et al. The potential of adjuvant against production of antistreptococcal immunoglobulin Y (IgY) in aquaculture. Indones J Vet Sci 2020;14. https://doi.org/10.21157/j.ked.hewan.v14i3.16911
[46] Arumugam M, Zhang Y, Huang Y, Perumal RK, Zhang T, Kong X, et al. Cell encapsulated biomaterials for translational medicine. Bioact Mater 2026; 56:402–54. https://doi.org/10.1016/j.bioactmat.2025.10.021
[47] Tayyab M, Islam W, Waqas W, Zhang Y. Probiotic–vaccine synergy in fish aquaculture: exploring microbiome-immune interactions for enhanced vaccine efficacy. Biology (Basel) 2025;14. https://doi.org/10.3390/biology14060629
[48] Gonciarz W, Balcerczak E, Brzeziński M, Jeleń A, Pietrzyk-Brzezińska AJ, Narayanan VHB, et al. Chitosan-based formulations for therapeutic applications: a recent overview. J Biomed Sci 2025; 32:62. https://doi.org/10.1186/s12929-025-01161-7
Downloads
Published
Issue
Section
License
Copyright (c) 2026 The Author(s)

This work is licensed under a Creative Commons Attribution 4.0 International License.




